1 / 2
0
Views
0
Downloads

0.0
0
0 Likes
Atomic Structure, Ions, and Isotopes Worksheet
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
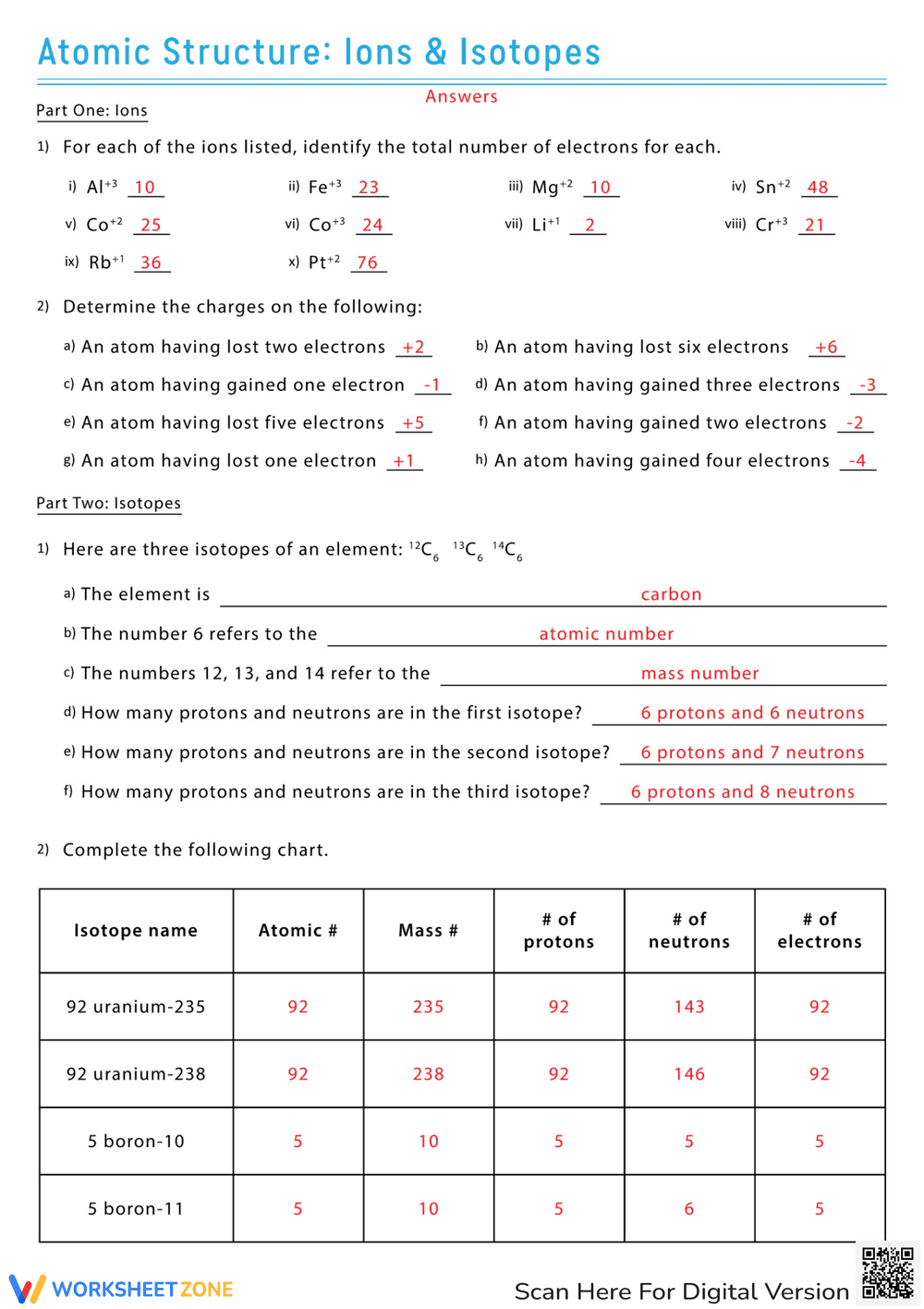
This is a chemistry worksheet focusing on atomic structure, specifically ions and isotopes. The worksheet is divided into two parts. The first part asks students to identify the number of electrons in various ions and to determine the charges on atoms that have lost or gained electrons. The second part presents questions about isotopes, including identifying an element given its isotopes, explaining what atomic numbers and mass numbers represent, calculating the number of protons and neutrons in different isotopes, and completing a chart with isotope names, atomic numbers, mass numbers, and the number of protons, neutrons, and electrons. Examples include Uranium-235, Uranium-238, Boron-10, and Boron-11.
Grade Level Suitability:
This worksheet is suitable for high school chemistry, specifically grades 9-12. It requires a basic understanding of atomic structure, ions, isotopes, atomic numbers, and mass numbers, which are typically covered in high school chemistry courses.
Why Use It:
This worksheet helps students reinforce their understanding of ions and isotopes by applying their knowledge to specific examples. It promotes critical thinking skills by requiring them to calculate the number of electrons, protons, and neutrons based on the given information. It also provides practice in identifying elements based on their isotopic composition.
How to Use It:
Students should first review the concepts of ions, isotopes, atomic number, and mass number. Then, they can work through the worksheet problems, showing their work where necessary. The first section requires calculating the number of electrons in each ion based on its charge. The second section requires applying knowledge of isotopes to determine the number of protons and neutrons and to complete the chart.
Target Users:
The target users are high school students studying chemistry. It is also suitable for teachers looking for supplementary materials to reinforce concepts related to atomic structure, ions, and isotopes.
This is a chemistry worksheet focusing on atomic structure, specifically ions and isotopes. The worksheet is divided into two parts. The first part asks students to identify the number of electrons in various ions and to determine the charges on atoms that have lost or gained electrons. The second part presents questions about isotopes, including identifying an element given its isotopes, explaining what atomic numbers and mass numbers represent, calculating the number of protons and neutrons in different isotopes, and completing a chart with isotope names, atomic numbers, mass numbers, and the number of protons, neutrons, and electrons. Examples include Uranium-235, Uranium-238, Boron-10, and Boron-11.
Grade Level Suitability:
This worksheet is suitable for high school chemistry, specifically grades 9-12. It requires a basic understanding of atomic structure, ions, isotopes, atomic numbers, and mass numbers, which are typically covered in high school chemistry courses.
Why Use It:
This worksheet helps students reinforce their understanding of ions and isotopes by applying their knowledge to specific examples. It promotes critical thinking skills by requiring them to calculate the number of electrons, protons, and neutrons based on the given information. It also provides practice in identifying elements based on their isotopic composition.
How to Use It:
Students should first review the concepts of ions, isotopes, atomic number, and mass number. Then, they can work through the worksheet problems, showing their work where necessary. The first section requires calculating the number of electrons in each ion based on its charge. The second section requires applying knowledge of isotopes to determine the number of protons and neutrons and to complete the chart.
Target Users:
The target users are high school students studying chemistry. It is also suitable for teachers looking for supplementary materials to reinforce concepts related to atomic structure, ions, and isotopes.




