1 / 3
0
Views
0
Downloads

0.0
0
0 Likes
Balancing Chemical Equations Worksheet - Chemistry Practice Act
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
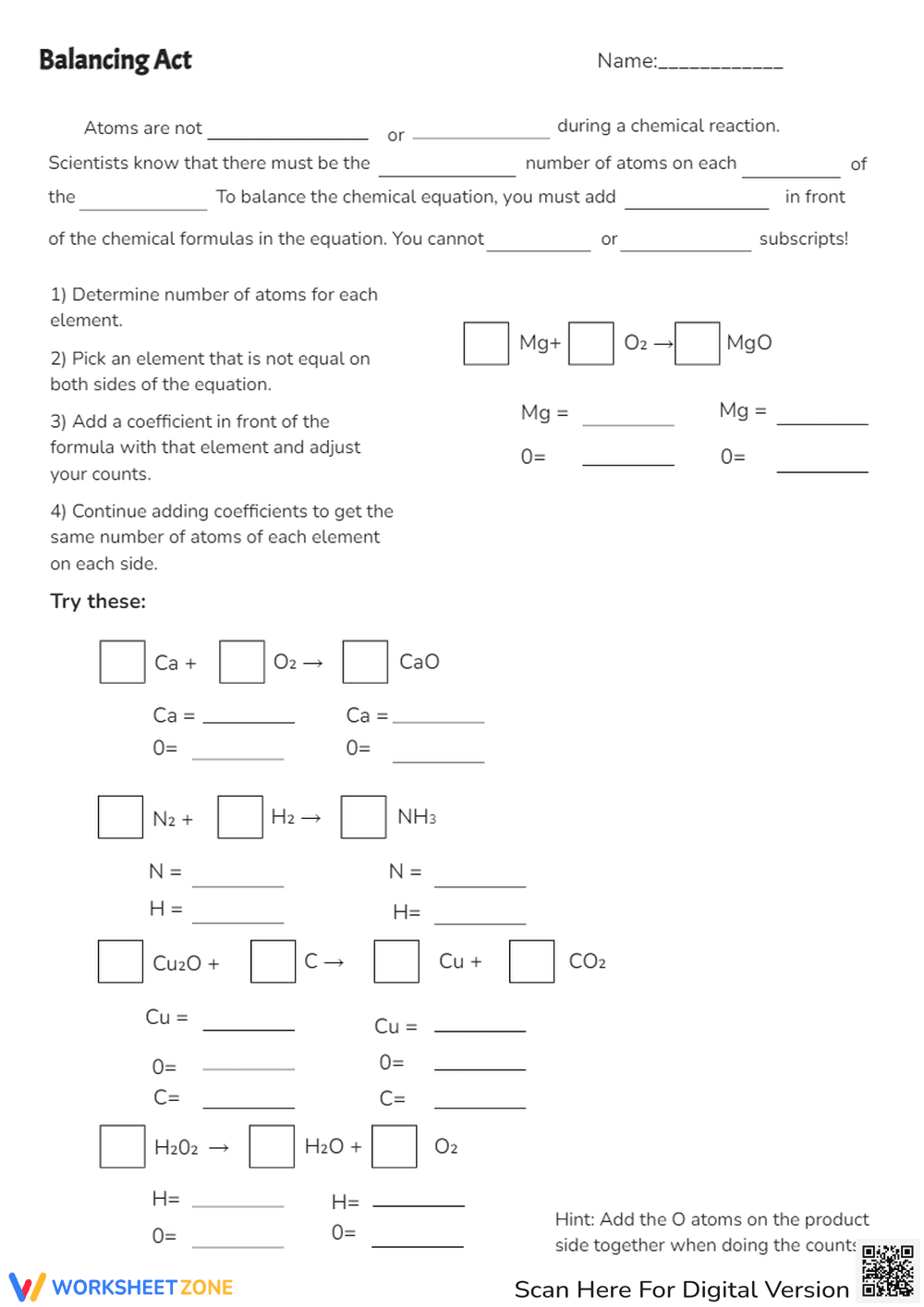
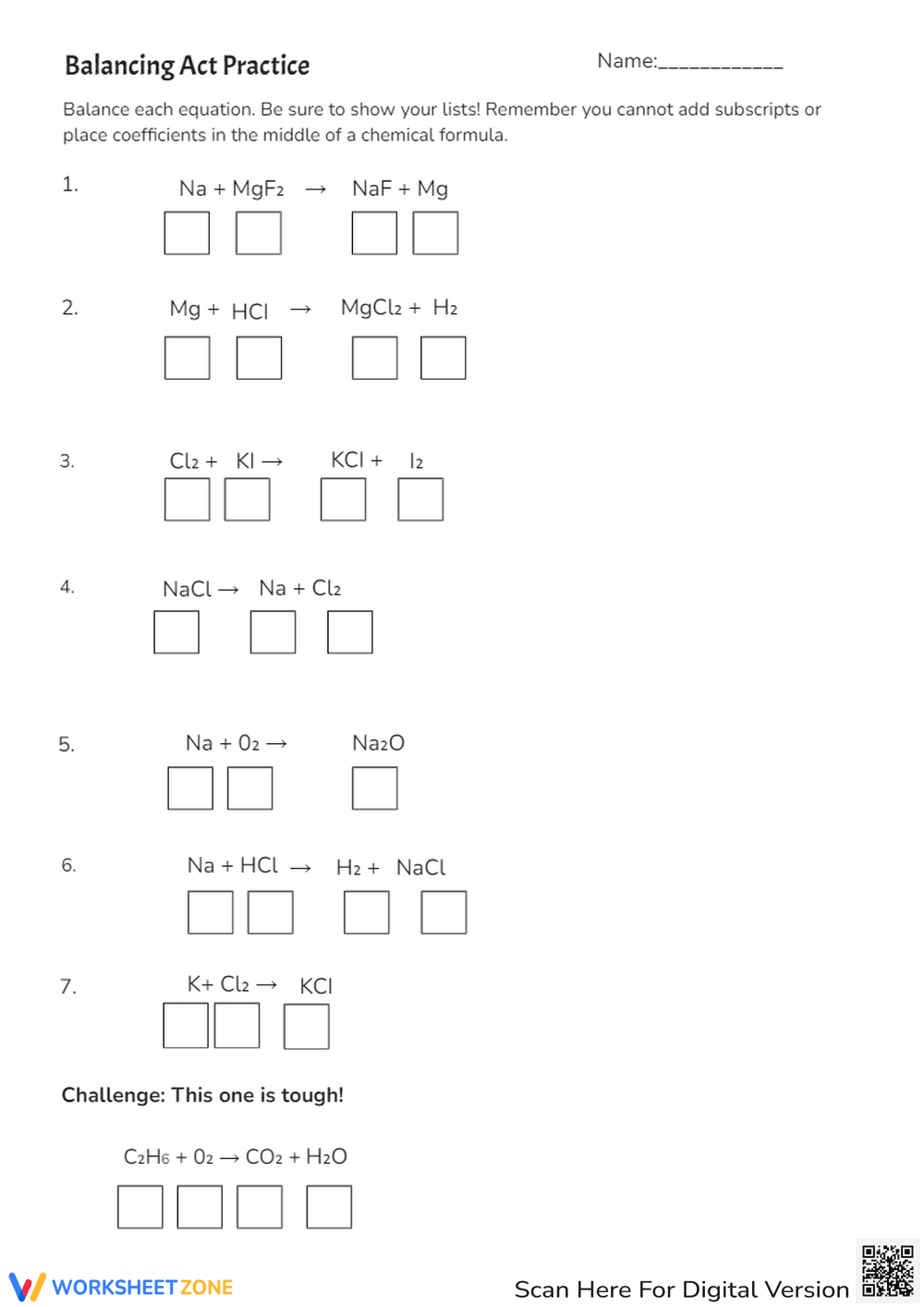
This is a chemistry worksheet titled 'Balancing Act' focused on balancing chemical equations. It provides instructions on how to balance equations by determining the number of atoms for each element, picking unequal elements, adding coefficients, and adjusting counts. The worksheet includes example problems with spaces to write coefficients and atom counts for equations like Mg + O2 -> MgO, Ca + O2 -> CaO, N2 + H2 -> NH3, CuO + C -> Cu + CO2, and H2O2 -> H2O + O2.
Grade Level Suitability:
This worksheet is suitable for high school grades (9-12), specifically for students learning about chemical reactions and stoichiometry in chemistry. The complexity of balancing equations requires a foundational understanding of chemical formulas and basic algebra.
Why Use It:
This worksheet helps students practice and master the skill of balancing chemical equations. It reinforces the law of conservation of mass by requiring students to ensure the same number of atoms of each element on both sides of the equation. It promotes problem-solving skills and a deeper understanding of chemical reactions.
How to Use It:
Students should follow the numbered steps provided to balance each chemical equation. First, determine the number of atoms of each element on both sides. Then, identify an element that is not balanced. Add a coefficient in front of the chemical formula containing that element and adjust the atom counts. Repeat until all elements are balanced. Write the coefficients in the provided boxes and the atom counts on the lines.
Target Users:
The target users are high school chemistry students who are learning about balancing chemical equations. It's also useful for teachers as a practice assignment or assessment tool to gauge student understanding of this concept.
This is a chemistry worksheet titled 'Balancing Act' focused on balancing chemical equations. It provides instructions on how to balance equations by determining the number of atoms for each element, picking unequal elements, adding coefficients, and adjusting counts. The worksheet includes example problems with spaces to write coefficients and atom counts for equations like Mg + O2 -> MgO, Ca + O2 -> CaO, N2 + H2 -> NH3, CuO + C -> Cu + CO2, and H2O2 -> H2O + O2.
Grade Level Suitability:
This worksheet is suitable for high school grades (9-12), specifically for students learning about chemical reactions and stoichiometry in chemistry. The complexity of balancing equations requires a foundational understanding of chemical formulas and basic algebra.
Why Use It:
This worksheet helps students practice and master the skill of balancing chemical equations. It reinforces the law of conservation of mass by requiring students to ensure the same number of atoms of each element on both sides of the equation. It promotes problem-solving skills and a deeper understanding of chemical reactions.
How to Use It:
Students should follow the numbered steps provided to balance each chemical equation. First, determine the number of atoms of each element on both sides. Then, identify an element that is not balanced. Add a coefficient in front of the chemical formula containing that element and adjust the atom counts. Repeat until all elements are balanced. Write the coefficients in the provided boxes and the atom counts on the lines.
Target Users:
The target users are high school chemistry students who are learning about balancing chemical equations. It's also useful for teachers as a practice assignment or assessment tool to gauge student understanding of this concept.




