1 / 2
0
Views
0
Downloads

0.0
0
0 Likes
Determining Stable Isotopes Practice Worksheet
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
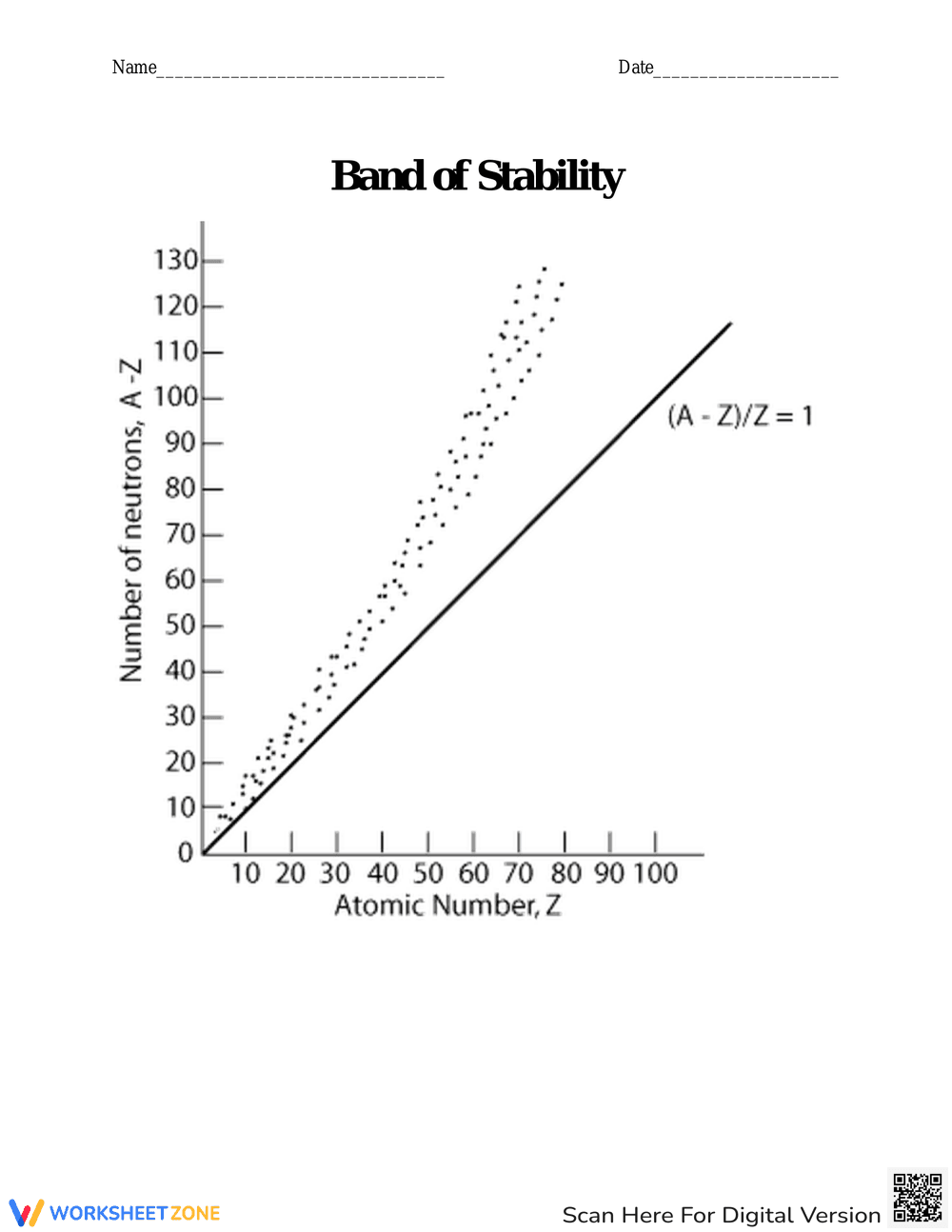
This is a chemistry worksheet titled 'Is an Isotope Stable?'. It presents a series of isotopes (48Cr, 86Br, 232Th, 39K, and 12N) and asks students to determine the number of protons and neutrons in each. Students must then determine if the isotope is stable or unstable and provide a reason for their answer. The worksheet also includes a brief explanation of the band of stability and a reminder that isotopes with atomic numbers greater than 83 are radioactive.
Grade Level Suitability:
This worksheet is suitable for high school students, specifically grades 9-12, in chemistry or physics courses. It requires an understanding of atomic structure, isotopes, and nuclear stability, concepts typically covered in these grades.
Why Use It:
This worksheet reinforces the concepts of isotopes, protons, neutrons, and nuclear stability. It helps students apply their knowledge to predict the stability of different isotopes and provides practice in justifying their answers. It also introduces the concept of the band of stability.
How to Use It:
Students should first determine the number of protons and neutrons for each isotope based on its atomic number and mass number. Then, using their understanding of the band of stability or other relevant rules, they should decide whether the isotope is stable or unstable. Finally, they should provide a brief explanation or justification for their answer.
Target Users:
The target users are high school students taking chemistry or physics courses, particularly those learning about atomic structure, isotopes, and nuclear chemistry. It's helpful for students needing extra practice in determining isotope stability.
This is a chemistry worksheet titled 'Is an Isotope Stable?'. It presents a series of isotopes (48Cr, 86Br, 232Th, 39K, and 12N) and asks students to determine the number of protons and neutrons in each. Students must then determine if the isotope is stable or unstable and provide a reason for their answer. The worksheet also includes a brief explanation of the band of stability and a reminder that isotopes with atomic numbers greater than 83 are radioactive.
Grade Level Suitability:
This worksheet is suitable for high school students, specifically grades 9-12, in chemistry or physics courses. It requires an understanding of atomic structure, isotopes, and nuclear stability, concepts typically covered in these grades.
Why Use It:
This worksheet reinforces the concepts of isotopes, protons, neutrons, and nuclear stability. It helps students apply their knowledge to predict the stability of different isotopes and provides practice in justifying their answers. It also introduces the concept of the band of stability.
How to Use It:
Students should first determine the number of protons and neutrons for each isotope based on its atomic number and mass number. Then, using their understanding of the band of stability or other relevant rules, they should decide whether the isotope is stable or unstable. Finally, they should provide a brief explanation or justification for their answer.
Target Users:
The target users are high school students taking chemistry or physics courses, particularly those learning about atomic structure, isotopes, and nuclear chemistry. It's helpful for students needing extra practice in determining isotope stability.




