1 / 5
0
Views
0
Downloads




0.0
0
0 Likes
Endothermic and Exothermic Worksheet 1: Chemistry
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
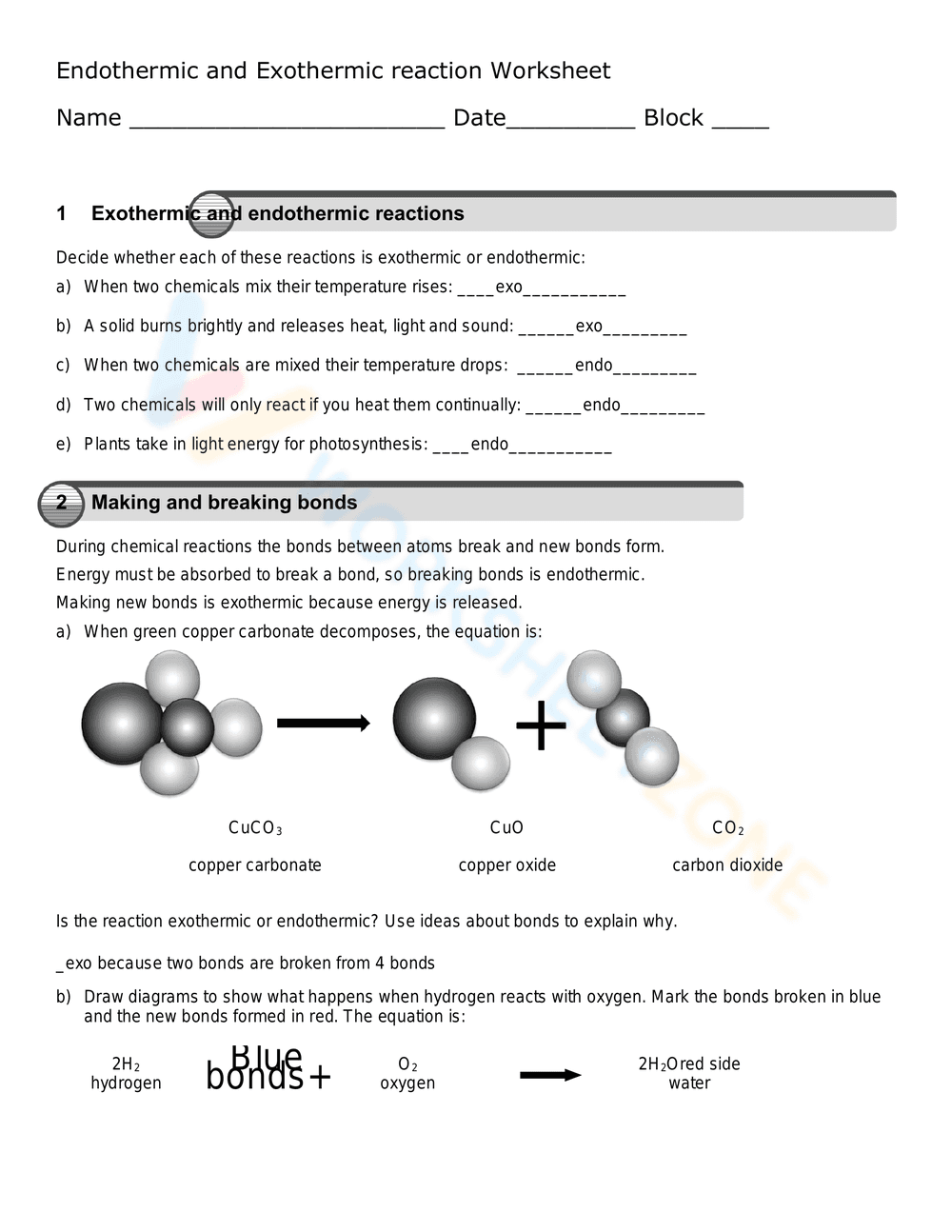
This is a chemistry worksheet focused on endothermic and exothermic reactions, and making/breaking bonds. It contains two sections. The first section asks students to identify whether reactions are exothermic or endothermic based on descriptions of chemical processes (e.g., temperature changes, burning). The second section presents a chemical reaction (decomposition of copper carbonate) with a diagram showing the molecules and asks students to determine if the reaction is endothermic or exothermic and explain why, and to draw diagrams illustrating the reaction between hydrogen and oxygen, marking broken and formed bonds.
Grade Level Suitability:
This worksheet is suitable for high school students, specifically grades 9-12, studying chemistry. The concepts of endothermic/exothermic reactions and bond energy are typically introduced at this level. The complexity of the chemical reactions and the need to draw diagrams require a basic understanding of chemical bonding and stoichiometry.
Why Use It:
This worksheet helps students understand the concepts of endothermic and exothermic reactions by applying them to real-world examples. It also reinforces the understanding of how energy is involved in breaking and forming chemical bonds. The diagramming activity promotes visual learning and a deeper comprehension of chemical reactions at the molecular level.
How to Use It:
Students should first read the descriptions of the chemical reactions in the first section and determine whether each reaction is endothermic or exothermic, writing their answers in the provided blanks. In the second section, they should analyze the provided reaction and explain whether it is endothermic or exothermic. Finally, they should draw diagrams to represent the reaction between hydrogen and oxygen, marking broken and formed bonds using different colors as instructed.
Target Users:
The target users are high school chemistry students, particularly those learning about thermochemistry and chemical bonding. It can also be used by teachers as a supplementary resource for classroom instruction or as a homework assignment to reinforce learning.
This is a chemistry worksheet focused on endothermic and exothermic reactions, and making/breaking bonds. It contains two sections. The first section asks students to identify whether reactions are exothermic or endothermic based on descriptions of chemical processes (e.g., temperature changes, burning). The second section presents a chemical reaction (decomposition of copper carbonate) with a diagram showing the molecules and asks students to determine if the reaction is endothermic or exothermic and explain why, and to draw diagrams illustrating the reaction between hydrogen and oxygen, marking broken and formed bonds.
Grade Level Suitability:
This worksheet is suitable for high school students, specifically grades 9-12, studying chemistry. The concepts of endothermic/exothermic reactions and bond energy are typically introduced at this level. The complexity of the chemical reactions and the need to draw diagrams require a basic understanding of chemical bonding and stoichiometry.
Why Use It:
This worksheet helps students understand the concepts of endothermic and exothermic reactions by applying them to real-world examples. It also reinforces the understanding of how energy is involved in breaking and forming chemical bonds. The diagramming activity promotes visual learning and a deeper comprehension of chemical reactions at the molecular level.
How to Use It:
Students should first read the descriptions of the chemical reactions in the first section and determine whether each reaction is endothermic or exothermic, writing their answers in the provided blanks. In the second section, they should analyze the provided reaction and explain whether it is endothermic or exothermic. Finally, they should draw diagrams to represent the reaction between hydrogen and oxygen, marking broken and formed bonds using different colors as instructed.
Target Users:
The target users are high school chemistry students, particularly those learning about thermochemistry and chemical bonding. It can also be used by teachers as a supplementary resource for classroom instruction or as a homework assignment to reinforce learning.




