1 / 5
0
Views
0
Downloads




0.0
0
0 Likes
Summary Intermolecular Forces Worksheet and Key
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
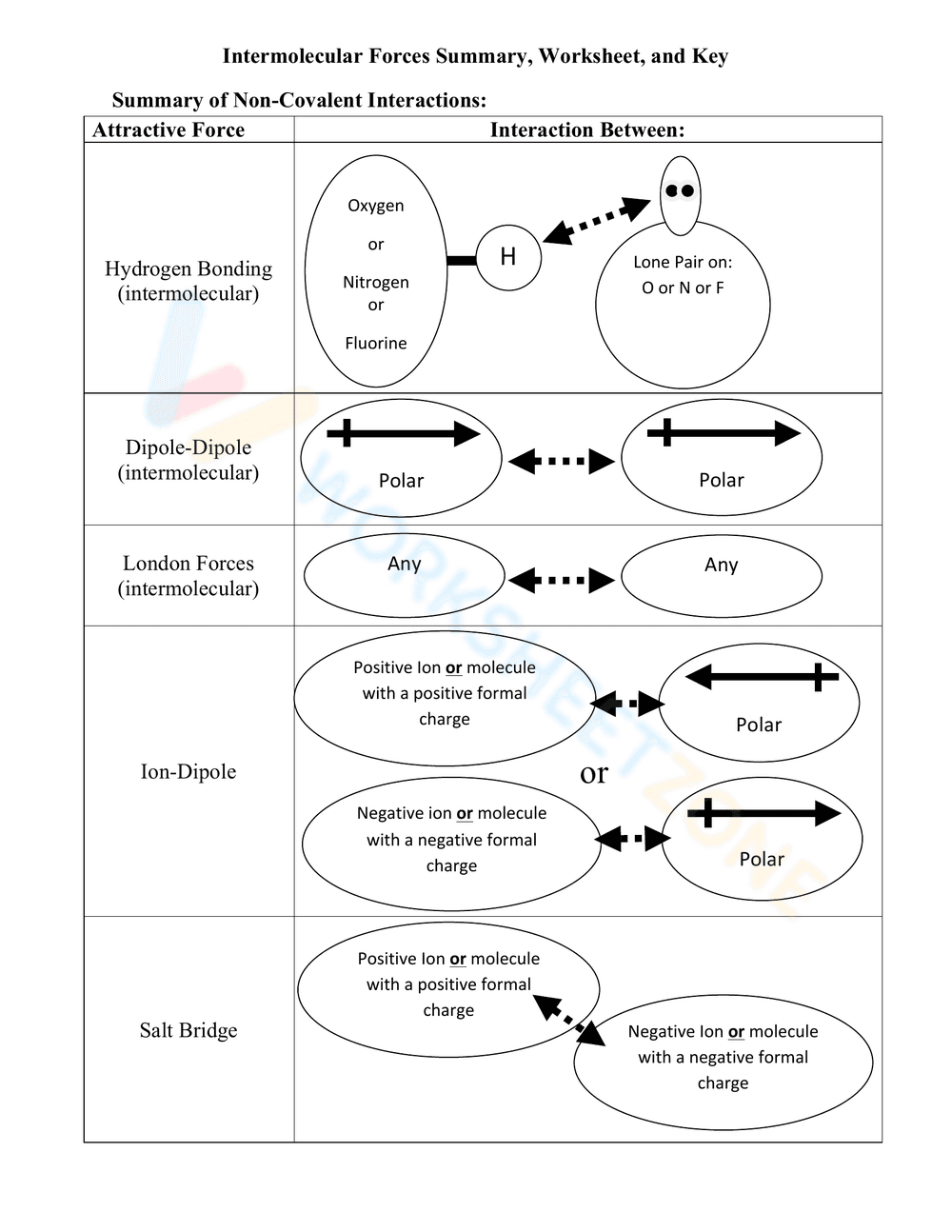
This is an educational worksheet summarizing non-covalent interactions, also known as intermolecular forces. It presents a table format with the 'Attractive Force' listed on the left and the corresponding 'Interaction Between' shown on the right. The worksheet includes visual representations and descriptions of Hydrogen Bonding, Dipole-Dipole, London Forces, Ion-Dipole, and Salt Bridge interactions.
Grade Level Suitability:
This worksheet is suitable for high school (Grades 11-12) and introductory college level chemistry courses. It requires understanding of basic chemistry concepts like polarity, ions, and molecular structure.
Why Use It:
This worksheet provides a concise visual summary of different types of intermolecular forces. It helps students to understand the interactions between molecules and the factors that influence these interactions. It also serves as a study guide or review tool.
How to Use It:
The worksheet can be used as a reference during lectures, as a review tool before tests, or as a guide for completing homework assignments. Students can use it to identify the types of molecules involved in each interaction and the nature of the attractive forces.
Target Users:
This worksheet is designed for students learning about intermolecular forces in chemistry. It's also useful for teachers as a teaching aid and for anyone who needs a quick reference guide to non-covalent interactions.
This is an educational worksheet summarizing non-covalent interactions, also known as intermolecular forces. It presents a table format with the 'Attractive Force' listed on the left and the corresponding 'Interaction Between' shown on the right. The worksheet includes visual representations and descriptions of Hydrogen Bonding, Dipole-Dipole, London Forces, Ion-Dipole, and Salt Bridge interactions.
Grade Level Suitability:
This worksheet is suitable for high school (Grades 11-12) and introductory college level chemistry courses. It requires understanding of basic chemistry concepts like polarity, ions, and molecular structure.
Why Use It:
This worksheet provides a concise visual summary of different types of intermolecular forces. It helps students to understand the interactions between molecules and the factors that influence these interactions. It also serves as a study guide or review tool.
How to Use It:
The worksheet can be used as a reference during lectures, as a review tool before tests, or as a guide for completing homework assignments. Students can use it to identify the types of molecules involved in each interaction and the nature of the attractive forces.
Target Users:
This worksheet is designed for students learning about intermolecular forces in chemistry. It's also useful for teachers as a teaching aid and for anyone who needs a quick reference guide to non-covalent interactions.




