0
Views
0
Downloads
0.0
0
0 Likes
Understanding pH and pOH
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
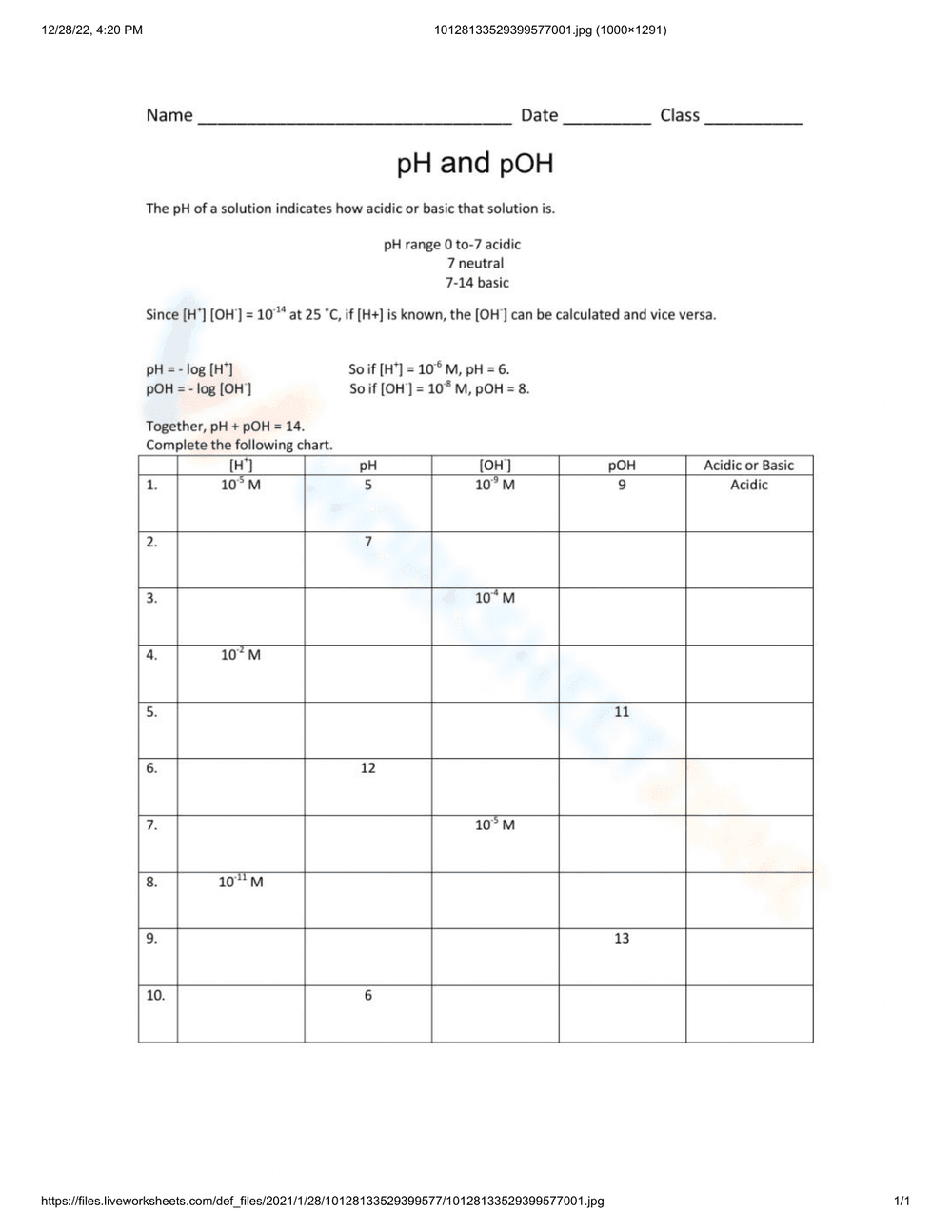
This is a chemistry worksheet focusing on pH and pOH calculations. It includes a brief explanation of the pH scale, formulas for calculating pH and pOH, and a chart to complete. The chart requires students to calculate pH, pOH, [H+], [OH-], and determine if a solution is acidic or basic based on given values.
Grade Level Suitability:
Suitable for high school chemistry, specifically grades 11-12. It requires an understanding of logarithms and chemical equilibrium concepts related to acids and bases.
Why Use It:
This worksheet helps students practice calculating pH and pOH, reinforces the relationship between [H+], [OH-], pH, and pOH, and provides practice in identifying acidic and basic solutions. It improves problem-solving skills and understanding of acid-base chemistry.
How to Use It:
Students should use the provided formulas (pH = -log[H+] and pOH = -log[OH-]) and the relationship pH + pOH = 14 to fill in the missing values in the chart. They need to determine whether each solution is acidic or basic based on its pH or pOH.
Target Users:
High school chemistry students learning about acids and bases, teachers looking for practice problems on pH and pOH calculations, and tutors assisting students with chemistry concepts.
This is a chemistry worksheet focusing on pH and pOH calculations. It includes a brief explanation of the pH scale, formulas for calculating pH and pOH, and a chart to complete. The chart requires students to calculate pH, pOH, [H+], [OH-], and determine if a solution is acidic or basic based on given values.
Grade Level Suitability:
Suitable for high school chemistry, specifically grades 11-12. It requires an understanding of logarithms and chemical equilibrium concepts related to acids and bases.
Why Use It:
This worksheet helps students practice calculating pH and pOH, reinforces the relationship between [H+], [OH-], pH, and pOH, and provides practice in identifying acidic and basic solutions. It improves problem-solving skills and understanding of acid-base chemistry.
How to Use It:
Students should use the provided formulas (pH = -log[H+] and pOH = -log[OH-]) and the relationship pH + pOH = 14 to fill in the missing values in the chart. They need to determine whether each solution is acidic or basic based on its pH or pOH.
Target Users:
High school chemistry students learning about acids and bases, teachers looking for practice problems on pH and pOH calculations, and tutors assisting students with chemistry concepts.




