0
Views
0
Downloads
0.0
0
0 Likes
Subatomic Particles: Worksheet for Learning
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
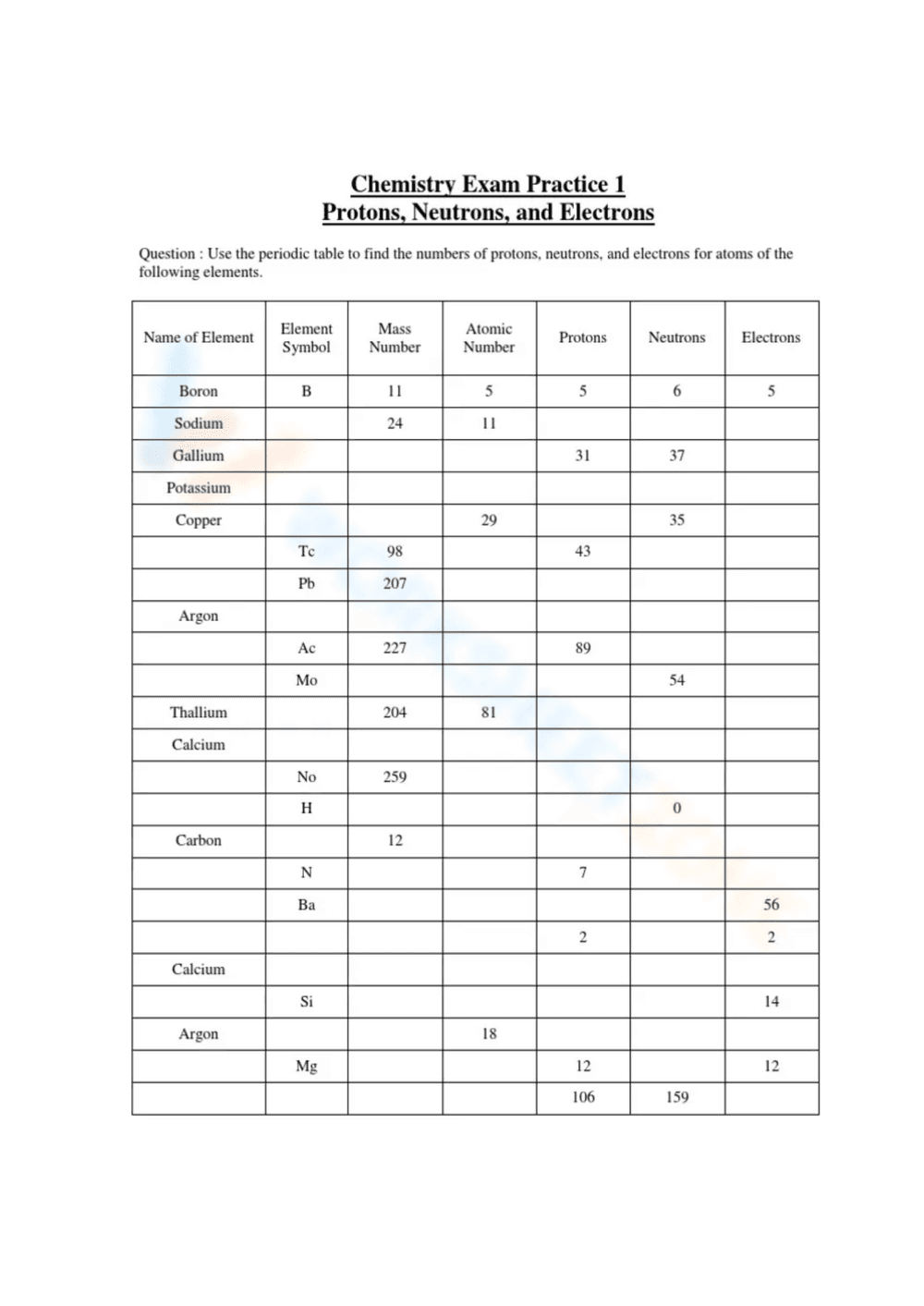
This is a chemistry worksheet titled 'Chemistry Exam Practice 1: Protons, Neutrons, and Electrons.' It presents a table where students are asked to fill in the number of protons, neutrons, and electrons for various elements, given the element's name, symbol, mass number, and atomic number. Some of the cells are pre-filled, requiring the student to deduce the missing information. Elements listed include Boron, Sodium, Gallium, Potassium, Copper, Tellurium, Lead, Argon, Actinium, Molybdenum, Thallium, Calcium, Nobelium, Hydrogen, Carbon, Nitrogen, Barium, Silicon, and Magnesium.
Grade Level Suitability:
This worksheet is most suitable for high school chemistry students (Grades 9-12). It requires an understanding of atomic structure, the periodic table, and the relationship between atomic number, mass number, and the number of protons, neutrons, and electrons.
Why Use It:
This worksheet reinforces understanding of fundamental chemistry concepts related to atomic structure. It helps students practice using the periodic table to determine the number of protons, neutrons, and electrons in an atom. It also serves as a good exam preparation tool.
How to Use It:
Students should use a periodic table to find the atomic number and mass number for each element listed. They can then use the provided information to calculate the number of protons, neutrons, and electrons. The completed table will demonstrate their understanding of atomic structure.
Target Users:
The target users are high school chemistry students, teachers, and tutors looking for practice materials on atomic structure and the periodic table. It can also be used for exam review and reinforcement of key concepts.
This is a chemistry worksheet titled 'Chemistry Exam Practice 1: Protons, Neutrons, and Electrons.' It presents a table where students are asked to fill in the number of protons, neutrons, and electrons for various elements, given the element's name, symbol, mass number, and atomic number. Some of the cells are pre-filled, requiring the student to deduce the missing information. Elements listed include Boron, Sodium, Gallium, Potassium, Copper, Tellurium, Lead, Argon, Actinium, Molybdenum, Thallium, Calcium, Nobelium, Hydrogen, Carbon, Nitrogen, Barium, Silicon, and Magnesium.
Grade Level Suitability:
This worksheet is most suitable for high school chemistry students (Grades 9-12). It requires an understanding of atomic structure, the periodic table, and the relationship between atomic number, mass number, and the number of protons, neutrons, and electrons.
Why Use It:
This worksheet reinforces understanding of fundamental chemistry concepts related to atomic structure. It helps students practice using the periodic table to determine the number of protons, neutrons, and electrons in an atom. It also serves as a good exam preparation tool.
How to Use It:
Students should use a periodic table to find the atomic number and mass number for each element listed. They can then use the provided information to calculate the number of protons, neutrons, and electrons. The completed table will demonstrate their understanding of atomic structure.
Target Users:
The target users are high school chemistry students, teachers, and tutors looking for practice materials on atomic structure and the periodic table. It can also be used for exam review and reinforcement of key concepts.




