0
Views
0
Downloads
0.0
0
0 Likes
Understanding the Bohr Model of the Atom
0 Views
0 Downloads
Paste this activity's link or code into your existing LMS (Google Classroom, Canvas, Teams, Schoology, Moodle, etc.).
Students can open and work on the activity right away, with no student login required.
You'll still be able to track student progress and results from your teacher account.
Information
Description
What It Is:
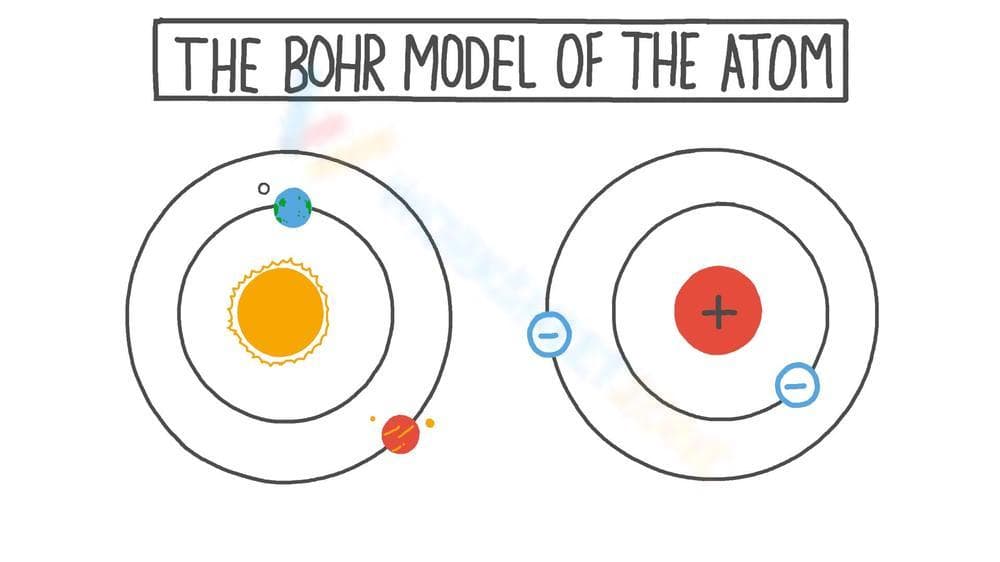
This worksheet depicts the Bohr model of the atom. It shows two atoms, each with a nucleus at the center and electrons orbiting in defined paths or energy levels. One atom shows an electron transitioning between energy levels, indicated by a colored arrow. The other atom illustrates electrons orbiting the nucleus, labeled with negative signs. The nucleus in the second atom is labeled with a positive sign.
Grade Level Suitability:
Suitable for grades 8-12. The Bohr model is typically introduced in middle school or high school science classes when students are learning about atomic structure and basic chemistry concepts.
Why Use It:
This worksheet helps students visualize the Bohr model of the atom and understand the arrangement of electrons around the nucleus. It illustrates the concept of electrons existing in specific energy levels and the transition of electrons between these levels, which is fundamental to understanding atomic behavior and chemical bonding.
How to Use It:
Use this as a visual aid to explain the Bohr model. Students can label the parts of the atom (nucleus, electrons, energy levels). The image can also be used as a starting point for a discussion about the limitations of the Bohr model and the development of more advanced atomic models.
Target Users:
Middle and high school science students learning about atomic structure, chemistry teachers, and homeschooling parents teaching basic chemistry concepts.
This worksheet depicts the Bohr model of the atom. It shows two atoms, each with a nucleus at the center and electrons orbiting in defined paths or energy levels. One atom shows an electron transitioning between energy levels, indicated by a colored arrow. The other atom illustrates electrons orbiting the nucleus, labeled with negative signs. The nucleus in the second atom is labeled with a positive sign.
Grade Level Suitability:
Suitable for grades 8-12. The Bohr model is typically introduced in middle school or high school science classes when students are learning about atomic structure and basic chemistry concepts.
Why Use It:
This worksheet helps students visualize the Bohr model of the atom and understand the arrangement of electrons around the nucleus. It illustrates the concept of electrons existing in specific energy levels and the transition of electrons between these levels, which is fundamental to understanding atomic behavior and chemical bonding.
How to Use It:
Use this as a visual aid to explain the Bohr model. Students can label the parts of the atom (nucleus, electrons, energy levels). The image can also be used as a starting point for a discussion about the limitations of the Bohr model and the development of more advanced atomic models.
Target Users:
Middle and high school science students learning about atomic structure, chemistry teachers, and homeschooling parents teaching basic chemistry concepts.




